The American Chemical Society
“I hardly know which we ought most to rejoice at- the progress that has been made in natural knowledge or the progress that is to be made.” -Davy.

The American Chemical Society (ACS) is the world’s largest scientific society with around 157,000 (I am one of these members!) It is also one of the world’s leading sources of scientific information. The ACS is at the forefront of the worldwide chemistry enterprise.
“We strive to be a force for good.”
The menu I chose as my inspiration for this project was one used at the 25th anniversary banquet for the American Chemical Society on April 13, 1901 at Hotel Savoy.
It was the toasts preceding the menu that first caught my attention; major figures within ACS spoke of science and progress. Immediately after seeing this, I knew what direction I wanted to take this assignment. Being a chemistry major, it is interesting to me to see what chemicals make up our foods. I always think about how these additives got to be there and how they are helping and possibly hurting the consumer. I wanted to delve into the history of food chemistry, especially food safety, and compare it to food chemistry now. I wanted to attempt to bake something from the menu and compare the chemicals used then to the chemicals used now.
Has chemistry helped American food progress or has it set food back?
Before the Civil War, people grew and raised their food on their farms; chemical additives were obsolete. At the turn of the twenty century, this changed. Thousands of rural people flocked to the cities to work in factories. Now, there was a need to have food preserved and packaged by someone else.

The industrial revolution scaled food adulteration to extraordinary levels (some common food adulterants are shown in the table below.) Food purity was not a concern; cheap and quick methods of preserving foods was the name of the game. Food chemistry was basically nonexistent.
At this same time, advances were being made in analytical chemistry. This was key in developing food protection legislation.
Below are key events that have led better regulations:
Late 17th century: British chemist Robert Boyle began developing new assays for food adulterants.
1820: German chemist Frederick Accum published Treatise on Adulteration of Food, and Culinary Poisons, the first work to raise awareness in this area.
1850: British physician Arthur Hassall began investigating food safety in England.
**Work by Hassall and Accum was the motivation behind food reform in the United States.
1862: Congress creates the Department of Agriculture, Division of Chemistry.
1902: Harvey Wiley begins to study the health effects of chemicals in food by feeding healthy males common food adulterants at the time. Although these experiments were extremely flawed, they influenced policy change.

1906: The Pure Food and Drugs Act (PFDA) was signed by Teddy along with the Meat Inspection Act inspired by Upton Sinclair’s The Jungle.
The PFDA banned false and misleading statements on packaging.
The PFDA was assigned to the USDA Bureau of Chemistry, later to be known as the FDA.

1920: Food adulteration remains an issue due to holes in PFDA.
1927: PFDA was enforced until now. The Food, Drug, and Insecticide Administration was formed.
1931: The Food, Drug, and Insecticide Administration was renamed the Food and Drug Administration (FDA).
1933: The FDA recommends a complete revision of PFDA.
1938: Congress passes the Food Drug and Cosmetic Act (FDCA).
It required that certain components be identifiable and mandated factory inspections. It required that coloring be approved before use in foods. The FDCA established tolerance levels for certain toxins.
Since these advancements, food chemistry has continued to develop.

Homemade Petit Fours
As someone who prides herself on the amount of months I can go without going to the grocery store (my record is 3 months), it is pretty clear I do not enjoy cooking. I look at it as a waste of time…plus I’ll have to clean up after. I’ll just pick up Subway or eat at my favorite restaurant in New Orleans, the Orleans Room. So, when I was first considering what my creative take on this menu analysis would be, in no way did I think I’d end up in the kitchen. At that point, I had my cooking blinders on. Baking never even crossed my mind.
I used to bake at least once a week before I attended college. It was one of my favorite hobbies. To this day, I still am hoping to eventually open my own bakery that will be filled with the most beautiful desserts you have ever seen. I quit baking because I had no time and the kitchens in Biever Hall were not exactly areas I felt confident that I wouldn’t get Ebola or some other deadly virus. Also, I was trying my hardest to combat the Freshmen 15. So then, I decided that this would be the perfect opportunity for me to break out my whisk once more. I would attempt to bake, from scratch, a dessert from the menu. Desserts are not something that we have really discussed much in this class which is interesting to me. Really though, why are desserts always an afterthought (literally)? P.S. I am the person who eats desserts first.
Let’s get baking!
So by this point in my night, it is 12:30am. I decided to start baking a pound cake at 12:30am…I will try my hardest to recount all difficulties and triumphs I encountered, but this night was a little fuzzy.
First, I started by making the pound cake. (I read online that pound cakes were best for petit fours since the dense cake would provide the stability needed.) I went to Winne Dixie to gather all of the supplies. 40 minutes and 40 dollars later…
- 3 sticks (339 g) unsalted butter, softened
- 3 cups (600 g) sugar
- 5 large eggs
- 3 cups (375 g) all purpose flour, (not self-rising)
- 1/2 teaspoon (2 g) baking powder
- 1 cup (235 g) whole milk
- 2 teaspoons (8 g) vanilla
- 1 teaspoon (4 g) lemon extract (optional)
**I do organic chemistry research. I very much enjoy it and I think I do so because I find it very similar to baking. There is an exact science to baking yet so much room for discovery. This is what I like about baking. I like changing the recipes ever so slightly (not that it even makes a difference) because it makes me feel like I am a great baker. I like throwing random things into a bowl because I think it will taste good in the end. So, this is why you will often see me doing things slightly differently than what I am told will yield a good cake.
First, I preheated the oven to 325 degrees, although the recipe I was following suggested 350. I then creamed the butter with a mixer. Luckily, my roommate had just gotten one or this whole process would have been a lot more painful. (The electric standing mixer was invented in 1908, so there was definitely some manual labor involved in the original baking of these petit fours.) I gradually added the sugar and beat the mixture until light and fluffy. I used a really low setting through my baking adventure as I was baking in the middle of the night and wanted to keep the noise to a minimum. I then added the eggs one by one. I added the vanilla and milk. Instead of opting for lemon extract, I added a shit ton of almond extract, strictly a personal preference. In a separate bowl, I whisked together the flour and baking powder.The recipe suggested I add the flour mixture alternately with the milk to the butter, sugar, egg mixture and to begin and end with the dry ingredients. This seemed annoying to me so I just threw it all together. Now it was done! It smelt good and tasted good. I buttered and floured two 13×9 pans and filled them with batter. Wow this is a lot of batter…I am not planning on making this many petit fours!! Then the recipe tells me to bake my cakes for one hour and ten minutes. ONE HOUR AND TEN MINUTES?? I put the cake in at 1:20am…it’s going to be a long night.
While the cake was in the oven for what seemed like a year, I started making the icing. At this point I did not feel like following any sort of recipe. I tried to recall my favorite icing from my childhood and how my mom used to make it. My mom was not awake at this point for me to ask her, so I just guessed…based on the taste, probably not my best idea. I started by creaming a stick of Crisco, yum. I then added as much powdered sugar and milk as my heart saw fit. I added random amounts of vanilla and almond extract. Something was missing…it tasted funny. Crap, my mom usually adds butter extract. I do not have butter extract. What even is butter extract? So I decide to throw away half of the icing I made and the use other half as a base for the new one I would attempt to make. (I was using small bowls so this was necessary.) I added a stick of butter because this has to be a little similar to the butter extract right? After adding rounds of butter, milk, and powdered sugar, I had the consistency I wanted. It still didn’t taste the greatest, but it’ll do.
I take the cakes out probably ten minutes too soon, but I am tired and generally impatient. I am also too impatient to let them cool. I immediately plop them out of the pans, hoping for a quicker cooling process. I find a random object in my pantry that will allow me to trace perfect squares onto my cake. I cut the squares and put them in the fridge. Admittedly, these squares were much too large. I began to ice the cakes which proved itself to me a challenge. Icing little baby cakes is annoying and hard to do. I have small hands but not small enough for this task. After what I thought was meticulously icing these cakes (they were kind of ugly), I put them in the fridge for some time to harden up a little while I made the glaze to go on top.
Making the glaze was an interesting experience. The recipe called for:
- 9 cups sifted confectioners’ sugar (sift then measure) (945 g)
- 1/2 cup (164 g) light corn syrup
- 1/2 cup water (118g)
- 1 teaspoon clear vanilla extract (4g)
- 1/2 teaspoon almond extract (optional)
I was supposed to in a heatproof bowl set over (but not touching) simmering water, mix sugar, corn syrup, 1/2 cup water, and flavoring together until warm and smooth. I was supposed to use a candy thermometer to heat up the mixture to 115 degrees. I do not own a candy thermometer so everything I was about to do was going to be a guess. Also, I do not own a double boiler. I decide a use a pot with water and a glass cereal bowl that seemed like it would do the trick. It did not. Nothing worked. Maybe I burned it? It was just like a hard tar ball. So naturally I just added a lot of water until a glaze consistency was reached, and I called this my glaze. I poured the glaze over my five petit fours. (Yes, I literally only made 5 because I couldn’t even imagine the labor it would take to make much more than that.) The glaze never did harden; the petit fours were a little wet. It is now 4am. I want to go to sleep. The petit fours are ugly, arguably disgusting, and very very dense. I will never look at the petit four the same.



Plot Twist…

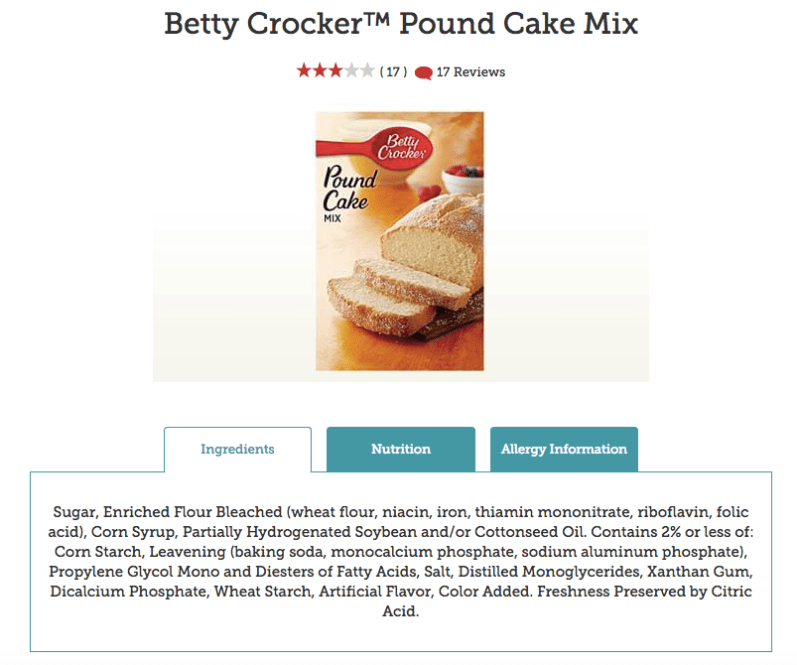
The direction I thought this paper would go in (prior to research) was much different than what I have now. I planned on researching the history of food chemistry and finding that we have been pumping our food with more and more chemicals as time goes on. I expected the food that would have been made in 1901 on this menu to be pure. I wanted to bake petit fours how they would have been made at this dinner and compare their ingredients to the ingredients in the box cake mix and canned icing.
My assumptions were very wrong. The food at the time of this dinner was in no way “pure.” This time period is home to arguably the worst food chemistry the United States has ever seen. It was during this time that adulterated foods were at an all time high. It is very likely that items on this menu such as milk, wine and beer (although not on the menu, I am assuming because of the toasts), vinegar, pepper, bread, butter, maple syrup, olive oil, and honey. Some of these items may have included adulterants such as red lead, dextrin, chalk, sulfuric acid, tin, lead, alum, borax, and lead chromate (Table 1.)
Looking at the ingredients in some of today’s grocery store cake mixes and canned icings may seem daunting. The list may seem long and unfamiliar, but the list of ingredients ensures these chemicals are present in less than 2% by mass (thanks toFDCA?). These chemicals are arguably safe and regulated by the FDA. Although these ingredients in these baking items may be safe, this does not mean our current times don’t face hazards of their own.
The FDA does not ensure that all foods we eat today are safe. For example, eighty percent of processed foods consumed in the United States are banned in most other countries. These include artificial food dyes, farmed salmon, brominated vegetable oil, olestra, azodicarbonamide, synthetic hormones, pesticides, antibiotics, BHA, BHT, arsenic, potassium bromate, and ractopamine meat among other harmful ingredients. Of recent concern is toxic chemical compounds formed by over heating food products.
Over heated meats such as barbecue and bacon have been noted to produce carcinogenic and genotoxic compounds. The AGFD of the ACS has been one of the leading organizations publishing information on this.
Acrylamide, a neurotoxin and carcinogen, has also been of great concern. In 2002 it was found in a wide range of potato chips, French fries, cereals, and roasted and baked potatoes. Acrylamide forms when you fry potatoes…this is the saddest thing I have ever read :(( It is speculated that in the United States and Canada diets consists of one-third of foods that are known to contain acrylamide. Since the ACS meeting in 2004, progress has been made in understanding the chemistry of acrylamide.
Many packaging contaminants enter our food as a biproduct of the food manufacturing process. BPA and PC are often found in plastic water bottles and baby bottles.
Agricultural and industrial contaminants may also affect the food supply. These include pesticides, fungicides, fertilizers, and veterinary drug residues.
This list of issues is not exhaustive; it is just to show that the United States still does face many issues within food chemistry and toxicology.
The future of food chemistry is most promising now. The development of new and increasingly sophisticated techniques allows for the authentication and analysis of foods. We are now more able than ever to detect chemical hazards, both more rapidly and at levels lower than previously possible.
I wonder if food chemistry was one of the topics discussed at the banquet…
Sources: Journal of Agricultural and Food Chemistry, “Chemical Food Safety Issues in the United States: Past, Present, and Future”



